The reason for the fall was mounting concerns over the approval of Medtronic’s most advanced insulin pump system – MiniMed 780G, which is already available in 38 countries and under review by the U.S. MDT stock fell over 6% in a single trading session on Dec 15, when the company made the FDA’s letter public. The agency is not pleased with how Medtronic handled its faulty MiniMed 600 insulin pumps, which were recalled in 2019, after more than thousands of complaints were reported over the span of three years. 
FDA’s warning letter to Medtronic for its Northridge facility in California citing inadequacy of medical devices quality system requirements. This underperformance can largely be attributed to the U.S. 
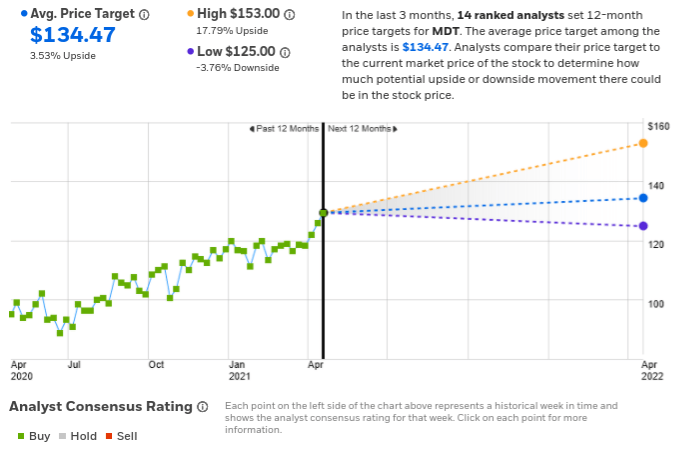
The stock price of Medtronic has seen a fall of 4% over the last month, underperforming the broader indices, with the S&P 500 rising 2% over the same period.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
